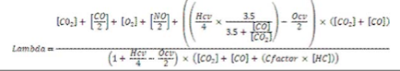I was manning the EEC stand at the recent Sandown Park MechanEx show, when a college tutor with six of his students asked me if I would show them the internal monolith of a catalytic converter and to explain to his students how it worked.
Lambda calculation – The Brettschneider Equation, general principles and methods
The Brettschneider equation is the de-facto standard method used to calculate the normalised air/fuel balance (Lambda) for domestic and international I&M inspection programs. It is taken from a paper written by Dr. Johannes Brettschneider, at Robert Bosch in 1979 and published in “Bosch technische Berichte”, Vol 6 (1979) N0. 4, Pgs 177-186.
In the paper, Dr. Brettschneider established a method to calculate Lambda (Balance of Oxygen to Fuel) by comparing the ratio of oxygen molecules to carbon and hydrogen molecules in the exhaust. The equation is a little complex, but is relatively easily calculated from the measured values of CO, CO2, unburned HC, and unconsumed O2 in the exhaust.As you see from the example on page 59, its a simple calculation!
As a result of the tutor’s theory I questioned several visitors using the same example, and was amazed at how many people had the same assumption regarding Lambda.
I know this can be misleading when looking at only the CO, HC and Lambda as required for an MoT report, but you need a four gas report to be able to make an in-depth emissions diagnosis, e.g. CO, CO2, HC, O2 and Lambda. These values are available from the majority of gas analysers on the market and can be printed on the MoT emissions report.
We all know that Lambda is a measurement of fuel/air (1 part fuel, 14.7 parts air). This measurement was calculated by Dr Johannes Brettschneider of Robert Bosch. Part of his paper is as follows:
CO @ .3, HC @ 203ppm and Lambda of 1.01, would show as a fail, as the CO and HC are just over the MoT limit of .2 & 200ppm respectively, however the Lambda reading is perfect.
Common mistake
A common error many technicians make when attempting to overcome the problem is changing the cat, as it has failed. Doing this could reduce the HC to between 100 > 160 PPM, and CO to <.2 which would now result in an MoT pass.
Unfortunately there is a fault and it needs correcting, otherwise it will result in the new catalytic converter failing again after just a few thousand miles.
How to overcome the problem
If you record all four gases in the example above, with the CO2 @ 14.2 and O2 @ .65 you would see that the O2 is too high. It should be <.2. The reason it is high is that air is entering the exhaust system between the front of the manifold and the first Lambda sensor.
Result
The Lambda sensor picks up the extra air, the ECU thinks that the mixture is lean and adds more fuel. Now there is extra fuel and air. If the fuel/air ratio is right, whatever the volumes it can still register a perfect Lambda.
Bad example
O2 @ .65, HC 203 PPM, CO2 @ 14.2, CO @ .3, Lambda of 1.01. Lambda is perfect. All other values are too high.
Good example
O2 @ .2, HC 0, CO2 @ 13.4, CO .1, Lambda of 1.00. All values are perfect. You can demonstrate this by going to www.smogsite.com/calculators. Scan down until you get to the Lambda calculator and enter some values.
As you increase the HC and the O2 you can still end up with a Lambda reading of 1.
The correct values are CO <.2 CO2> 13.5 HC <15 PPM O2 <.2 and all gas tests should be at 2500 rpm and at a temperature of 350 degrees.
I hope this explanation will show you that although Lambda is correct it does not mean that the air/fuel mixture is also right.